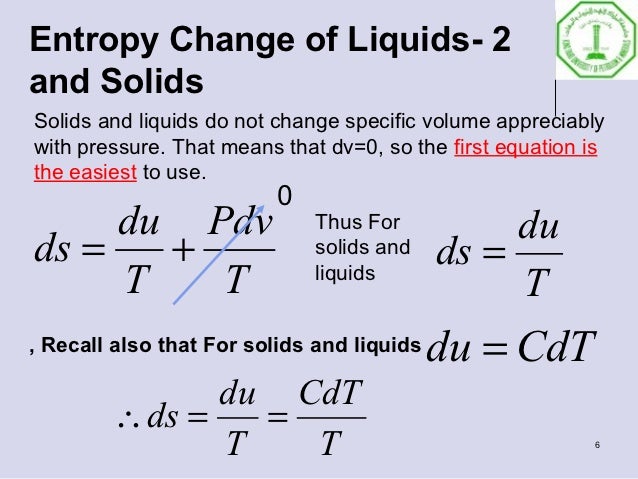
Δ G v a p = Δ H v a p − T v a p × Δ S v a p = 0 Īccording to Trouton's rule, the entropy of vaporization (at standard pressure) of most liquids is about 85 to 88 J mol -1 K -1. In a phase transition such as vaporization, both phases coexist in equilibrium, so the difference in Gibbs free energy is equal to zero. At standard pressure P o = 1 bar, the value is denoted as Δ S o vap and normally expressed in J mol -1 K -1. Solve the equation (specific heat at constant pressure Cp 4.1818 kJ/Kkg). Define final and initial temperature: Tf 20 C, Ti 100 C. This is always positive since the degree of disorder increases in the transition from a liquid in a relatively small volume to a vapor or gas occupying a much larger space. We will use the change in entropy formula: s Cp × ln (Tf / Ti), where Tf and Ti indicate the final and the initial temperature, respectively. C is 109.0 J K 1 mol 1 and the molar heat capacities at constant pressure of liquid water and water vapor are 75.3 J K 1 mol 1 and 33.6 J K 1 mol 1, respectively, in this range. The entropy of vaporization is the increase in entropy upon vaporization of a liquid. Calculate the standard entropy of vaporization of water at 85 C, given that its standard entropy of vaporization at 100. Eng.Template:Distinguish Template:Unreferenced stub Vapor-Liquid Equilibria and Interfacial Tensions of the System Ethanol + 2-Methoxy-2-methylpropane, Mejia, Andres Segura, Hugo Cartes, Marcela, Įnthalpies of Vaporization of Organic Compounds: A Critical Review and Data Compilation, Blackwell Scientific Publications, Oxford, 1985, 300. Vapor-Liquid Critical Properties of Elements and Compounds. SRD 156 – Clathrate Hydrate Physical Property Database SRSD 3 – Web Thermo Tables (WTT), professional edition SRSD 2 – Web Thermo Tables (WTT), "lite" edition 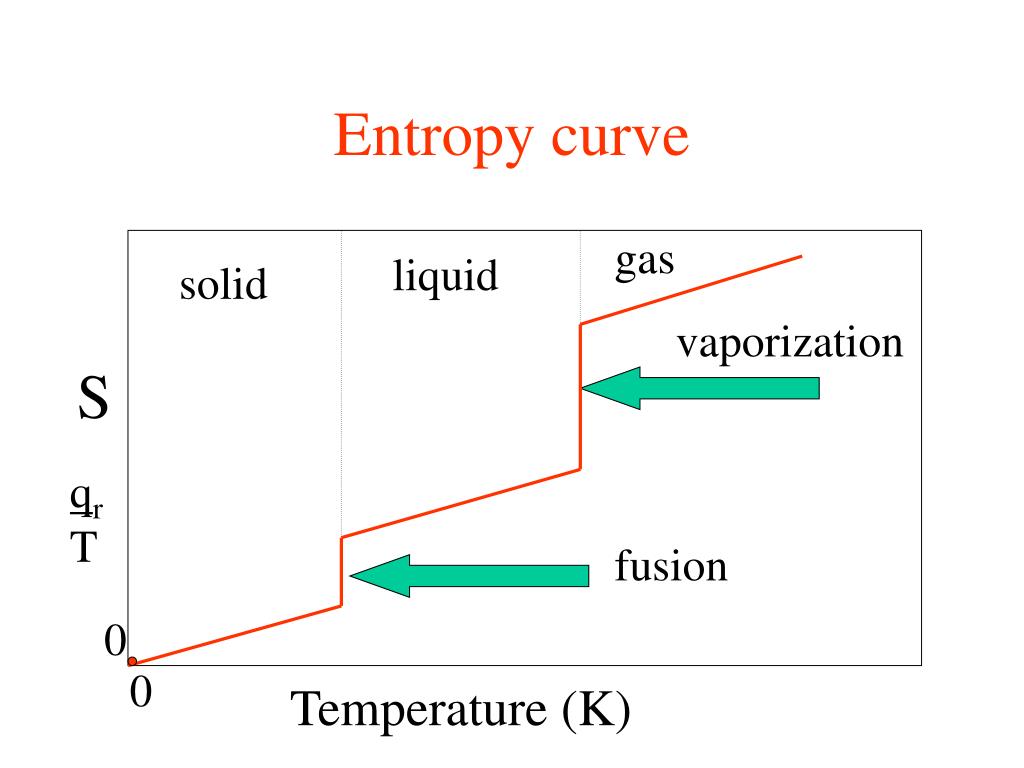
SRD 103b – Thermo Data Engine (TDE) for pure compounds, SRD 103a – Thermo Data Engine (TDE) for pure compounds. Calculate the entropy change for 1.0 mole of ice melting to form liquid at 273 K. The enthalpy of fusion for water is 6.01 kJ/mol. (TRC) data available from this site, much more physicalĪnd chemical property data is available from the The entropy change for a phase change at constant pressure is given by. In addition to the Thermodynamics Research Center Requires a JavaScript / HTML 5 canvas capable browser.Ĭoefficents calculated by NIST from author's data.Įnthalpy of phase transition ΔH trs (kJ/mol)Įntropy of phase transition ΔS trs (J/mol*K) ACĮnthalpy of vaporization (at saturation pressure) See also Ambrose, Counsell, et al., 1970. LouisĪverage of 138 out of 148 values Individual data pointsĪverage of 11 values Individual data pointsĪverage of 8 values Individual data pointsĪverage of 37 out of 38 values Individual data pointsĪverage of 18 out of 19 values Individual data pointsĪverage of 7 values Individual data pointsĪverage of 12 out of 13 values Individual data pointsīased on data from 293. Liebman, Students of Chem 202 (Introduction to the Literature of Chemistry), University of Missouri - St. TRC - Thermodynamics Research Center, NIST Boulder Laboratories, Chris Muzny directorīS - Robert L. Secretary of Commerce on behalf of the U.S.A. Your institution may already be a subscriber.įollow the links above to find out more about the dataīy the U.S. With the development of data collections included in The purpose of the fee is to recover costs associated NIST subscription sites provide data under theĭata Program, but require an annual fee to access. NIST / TRC Web Thermo Tables, professional edition (thermophysical and thermochemical data).NIST / TRC Web Thermo Tables, "lite" edition (thermophysical and thermochemical data).X-ray Photoelectron Spectroscopy Database, version 4.1.Computational Chemistry Comparison and Benchmark Database.Use this link for bookmarking this species This structure is also available as a 2d Mol file IUPAC Standard InChIKey: LFQSCWFLJHTTHZ-UHFFFAOYSA-N Copy.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
